This item was written in association with Dr Margarete Heck and her research group, The Wellcome Trust Centre for Cell Biology, Institute of Cell Biology, University of Edinburgh, UK.
KEY CONCEPTS:
CONCEPTS IN CONTEXT:
The division of a cell to produce two daughter cells is fundamental to most forms of life.
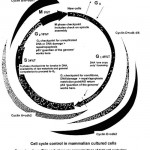
The ‘life cycle’ of a dividing eukaryotic non-embryonic cell starts with the cell triggered to enter the cell cycle and ends with the equal partitioning of the genetic material and cleavage of the cell during cytokinesis. The whole process is called the cell cycle and consists of four main phases.
Entry to the cycle is made in Gap 1 (G1) phase and this is followed in sequence by a DNA synthesis (S) phase, Gap 2 (G2) phase, and Mitosis (M). After mitosis (M) some cells enter the G1 phase of a new cell cycle whilst others may diverge at the start of G1 into a phase called Gap O (zero). Phases G1, S and G2 are often grouped and called ‘interphase’.
Cells in G-0 (zero) are quiescent and not dividing (hence zero), this may be permanent or temporary.
Mitosis (M phase) had been observed and described in some detail by the start of the 20th century, but it was not until about 50 years later that it was discovered that DNA synthesis took place as a separate process ahead of mitosis. Between mitosis (M) in a previous cell cycle and DNA synthesis (S) there was a time gap. This was designated Gap 1 (G1). The time gap between DNA synthesis (S) and mitosis (M) was designated Gap 2 (G2).
After cell division the daughter cells follow ONE of several pathways:
- cells that can divide again immediately enter the G1 phase of a new cell cycle.
- other cells enter G-0 phase. Some of them are quiescent for a time but then re-enter G1 phase.
- some specialist cells, for example nerve cells, do not divide again.
- other cells can be triggered by activities such as wound repair to enter G1 phase of the cell cycle to divide as required.
Mitosis is visually very dramatic but it only occupies about 5% of the total cell cycle time. Within the ‘big picture’ of the life cycle of a dividing cell, interphase (phases G1, S, and G2) account for the other 95% of cell cycle time. Research evidence shows interphase (once called resting phase!) operates in a beautifully ordered systematic way. It has also been shown to be more complicated than previously thought.
The time taken for a eukaryotic cell to divide varies widely with cell type and environment. Yeast cells take from 1.5 to 3 hours, intestinal epithelial cells about 12 hours and cells in culture about 22 hours. In different organisms and at different developmental times the details of the cell cycle vary. Embryonic cells in many organisms run a cycle that is shorter than similar cells in the adult. Cells of yeast and mammal show differences in cycle detail but the general mechanism of the cell cycle has been highly conserved over the years.
During the cell cycle cytoplasmic chemistry influences to a large extent the activities of the whole cell. At all other times we think in terms of the cell nucleus determining cytoplasmic activity.
KEY CONCEPTS:
- The cell cycle is a high quality cell production facility.
- An efficient production facility must have a controlled, co-ordinated and sequential system for producing products. Also needed is a system to monitor incoming raw materials, product processing, the processing environment and the facility to anticipate and deal with production problems.
- The eukaryotic cell is a ‘good manufacturer’ – most of the time! During the cell division cycle it has the biological equivalent of the following systems in place: Quality Assurance (QA), Quality Control (QC) and as part of QC, Internal Security (IS).
- At certain points in the cycle critical ‘reviews and decisions’ are made by QA and QC. These events are called ‘checkpoints’.
The cell cycle system of a eukaryotic organism includes:
- a biomolecular surveillance capability that integrates extracellular and intracellular signals and informs the cell’s QA and QC about: the need for a new cell; the availability of raw materials including a considerable amount of chemical energy; whether the microenvironment is suitable for cell division; and, the integrity of the genome and the fidelity of its replication (Mainly QA).
- a system that controls the cycling of the cycle and ensures the cell only enters the next phase when passage through the previous one is complete; also that the phases of the cell cycle are entered in the correct order. Satisfactory passage through all four phases and usually the various checkpoints of the cell cycle are critical for cell division (QC).
- a control system that initiates and terminates chemical reactions in the cell cycle and, with the use of a form of ‘licensing’ ensures that DNA is replicated once and once only during S phase (QC and IS).
- cell cycle arrest and delay functions (QC).
- DNA damage detection and repair facilities and a system that detects unreplicated DNA and, in mitosis, a system to ensure adequate spindle assembly and chromosome attachment (QA/QC), – otherwise known as “checkpoints”, and
- the capability and facility to trigger programmed cell death (apoptosis) (QC).
A cell is ‘cycled’ through each phase and from phase to phase by the action of proteins including specific cyclins and cyclin dependent kinases (cdks). Different cyclins and cdks rise and fall in activity during the cell cycle.
Sometimes faults go undetected (as in industry). Quality control and assurance systems can also fail. Quality control system failure is associated with cell and organ disease and probably as many as 50% of cancers (but this does not mean one single system failure alone will cause cancer).
KEY CONCEPTS: ENTRY LEVEL SUPPORTING INFORMATIONCells of eukaryotes normally only divide when instructed to do so. Chemicals called mitogens signal cells to start dividing.Cells competent to divide join the cell cycle in G1 phase and remain in this phase for a bit less than half of the total cell cycle time. This is the longest phase and microenvironmental conditions and signals received from other cells can shorten or lengthen G1.Early G1 phase
Recent work suggests that a licensing system for ‘one DNA copy only’ is set up early in G1 phase and expires only after DNA replication has started in S phase. During G1 phase only one set of the genome is present.
Late G1 phase
The main drivers of progression through the cell cycle are called protein kinases. There are several of these and each is a combination of a cyclin and a catalytic enzyme called a cyclin dependent kinase (cdk).
The different combinations operate in specific parts of the cell cycle and rise and fall in activity during the cycle. In so doing they contribute to the mechanism of phase entry and exit. The raised level of activity of the various cyclin and cdk combinations is terminated by proteolysis of cyclins after polyubiquitination. (Ubiquitins are a group of proteins that are covalently linked to proteins targeted for degradation. After molecular binding the target proteins, in this case cyclins, are degraded by proteolysis – protein loosening from Greek lusis, ‘loosening’)
The first cyclin combination (cyclin D and cdk 4 and 6) trips in about three quarters of the way through G1 phase to be joined later by the cyclin E and cdk 2 combination, both cyclins driving the cell into S phase
At about this time the cell passes through the ‘Restriction Point’ (in yeasts called START). This is a point of ‘no return’, the cell cycle equivalent of ‘Caesar crossing the Rubicon’. Once the cell passes this point it is restricted, there is no going back; the cell is committed to replicate in ‘S’ phase.
Checkpoints
As the cell progresses through the cycle, checkpoints are encountered. Although not absolutely essential for cell division they are well-established features of most cells.
Checkpoints in the cell cycle are not unlike those found at some border crossings where passports, papers and merchandise are checked. At the molecular level a cell cycle checkpoint consists of (1) sensor/detector (2) a signal sender and (3) a receiver/effector.
G1 checkpoint
The first of the ‘surveillance checkpoints’ is found towards the end of Gap1 (G1) phase and is the G1 DNA damage checkpoint. At this checkpoint and just ahead of it the DNA of the cell selected to divide is subjected to biomolecular surveillance for integrity. In humans DNA damage is ‘self-signalled’ mainly by the damaged DNA itself raising the level and activity of the protein products of certain genes, especially those of the tumour suppressor gene p53 from chromosome 17. Gene p53 is called the ‘guardian of the genome’ and is largely responsible for determining whether the cell should be admitted to the next synthesis (S) phase. If DNA is not too damaged a repair may be possible and the cell cycle arrested and slowed down until repairs are effected. Cell cycle ‘quality control’ may determine that the damage cannot be repaired. In this case ‘p53’ will trigger the programmed cell death (apoptosis) facility.
The division cycle of a cell with badly damaged DNA may end at this checkpoint.
Unfortunately, quality control in cells is not perfect. Very occasionally DNA damage is missed by QC and slightly changed DNA slips through the system. Sometimes this is because the damaged DNA does not trigger the checkpoint system. The quality control system may also be damaged itself. DNA of the p53 gene can be damaged by sunlight (u.v. radiation) and mutagenic chemicals including those from cigarette smoke and aflatoxin from, for example, mouldy peanuts.
At the G1 checkpoint in yeast cell cycle, a check is made to ensure cell size is correct for division, but cell size has been found not to be so critical a factor in some cells of higher eukaryotes
S (DNA synthesis) phase
S phase occupies about a third of total cell cycle time. It is here that under the ‘licensing’ system only one new copy of the cell’s DNA is synthesized. This includes acceptable alterations but also questionable ones not detected by QC because of system errors and breakdown. The failure of the ‘guardian of the genome’ p53 gene to operate due to damage within its own DNA is an example of this.
In S phase the double-stranded DNA unwinds into two component strands that serve as templates for the synthesis of a new strand on each. Newly formed units of the bases adenine (A), thymine (T), guanine (G) and cytsosine (C) are attached to compliment bases on the unwound DNA. One set of DNA is now produced for each of the two daughter cells. The process of DNA synthesis consumes a considerable amount of energy. (For details of this process see the textbooks listed on our website for example: Pollard, T. D. & Earnshaw, W. C., ‘Cell Biology’ 1st Edit. 2002. 2nd printing, with additions 2004. Publ: W. B. Saunders).
S phase checkpoints
There appear to be three types of checkpoint in S phase. Not surprisingly all respond in some way to problems with DNA replication. These problems range from a shortage of deoxyribonucleotides for making new DNA, to the presence of enzyme inhibitory chemicals and breaks in the DNA molecule. The cell cycle can arrest here if the DNA is unreplicated or in any way incomplete and not competent to proceed to phase G2.
If everything goes smoothly by the end of S phase the cell will contain two identical sets of its genome.
The cycle is driven through S phase and into G2 phase by the cyclin A and cdk 2 combination.
G2 (Gap 2) phase
G2 phase is generally shorter than that of G1. Much of this time is spent in developing and preparing organelles for dividing and sharing during cytokinesis at the end of mitosis. Cyclins A and B coupled to cdk 1 drive the cell through the end of S phase, through G2 phase and M phase This phase contains the G2 checkpoint. After G2 the cell is committed to division.
G2 Checkpoint
This DNA structure checkpoint is encountered towards the end of the G2 phase. The G2 checkpoint is very critical in that it has the ‘responsible’ function of providing a quality assurance check before the cell enters mitosis.
At the G2 checkpoint:
- monitoring takes place to ensure that there is no unreplicated DNA and that TWO identical sets of the genome are now present and intact.
- both sets of the genome are ‘proof-read’ with a high level of surveillance. A check is made for molecular damage within the DNA and this evidence will determine whether the genome will be retained, repaired or rejected.
- if repair of the damaged DNA is possible the cell cycle is arrested for as long as the repair takes. Cell cycles operate in ‘real time’ so delay can be accommodated within the system by QC delaying future events.
- badly damaged DNA will be identified and the p53 gene products will trigger the programmed death (apoptosis) programme.
- ‘Quality control’ will check the overall competence of the cell to enter mitosis. On the list of competences will be such items as “is the mechanism leading to the separation of the sister chromatids in place?”<
M phase (mitosis)
Mitosis presents the drama of division. An awesome and beautiful presentation, it occupies just 5% of the total cell cycle time, or roughly an hour in higher eukaryotic cultured cells. It is the shortest phase but the final production is the culmination of work done during the rest of the cell cycle.
Cyclin A and B coupled to cdk 1 drives the cell through mitosis (the student is referred to a textbook at the desired level for detailed information about mitosis). At the end of mitosis the sister chromatids, joined as pairs by cohesive ‘glue’ since they were replicated in S phase, are separated to form two new equivalent sets of chromosomes. This event is chromosome segregation.
Some organelles in the cytoplasm are disassembled into molecular units to be divided during division of the cytoplasm (kinesis), and then new ones constructed in the daughter cells. Other cytoplasmic inclusions are shared (not always evenly) between the daughter cells.
M phase (mitosis) checkpoints
The working of one checkpoint within mitosis, the metaphase checkpoint, is well established. There may be more checkpoints but further work is needed to establish their existence.
Metaphase checkpoint
Also called ‘spindle assembly or kinetochore attachment checkpoint’ it operates during metaphase and before the cell enters anaphase. It checks for misaligned chromosomes and also that microtubules are attached to kinetochores – a very critical mechanism. The start of anaphase is delayed until all the chromosomes are aligned and appropriately attached.
When telophase is complete cytokinesis – (the division of cytoplasm) takes place. After this the two daughter cells will be directed to G-0 (zero) or G1 phase.
CONCEPT MESSAGES:
- Research evidence shows very clearly that cell division in eukaryotes should now be viewed as an active multiphase process called the CELL CYCLE. The process is highly conserved and covers all the phases of preparation as well as the actual cell division. Satisfactory passage through the phases and checkpoints of the cycle are critical for cell division to take place.
- Mitosis whilst obviously very important and dramatic should be viewed as part of the ‘Big Picture’ of cell division. Mitosis occupies just 5% of the total cell cycle time.
- Working within the cell cycle are various forms of molecular quality assurance and control. These controls operate to assist the cell division programme and prevent damage to the genome.
CHALLENGE YOUR CRITICAL THINKING:
- Cancer is about cells dividing in an unregulated way. The nature of the cell cycle control system lends itself to the design of anti-cancer drugs that disrupt the cell cycle and initiate programmed cell death.
Consider the total cell cycle. Select two or more strategic sites as possible targets for the action of anti-cancer drugs? Think through in terms of cell and molecular biology what you would want the drug to do in order to interfere with the cell cycle. - If DNA replication and cell division were always perfect in every case what would be the implications for evolution?
SELECTED WEBSITES:
http://nobelprize.org
Click on Cell Cycle game in the education panel.
http://www.cancerresearchuk.org
Click on ‘Science & Research’ in the text. (Then select Annual Reports especially LRI 2002 & Sc.Yearbook 02/03)
http://www.cellsalive.com
Click on ‘cell cycle’ (select ‘checkpoints for animation’).
Grateful thanks are due to Margarete Heck for her valuable contribution.
Diagram ‘Cell cycle control in mammalian cultured cells’