(microtubules, and, forthcoming: intermediate filaments, actin filaments, and the motor proteins: kinesin, dynein and myosin)
To view a research level microscope image, interpreted using CIMR GridPoint technology, CLICK HERE
Quick look:The cytoskeleton is the overall name given to protein filaments and motor proteins (also called molecular motors) in the cell. These protein filaments form an enormous three dimensional (3D) meshwork. Filaments can be cross linked to other similar filaments, and to membranes, by means of accessory proteins. This inter-linking greatly increases rigidity. Some filaments are used as trackways for motor proteins to transport cargoes.
If you have heard about people who have been treated for cancer with the drugs taxol or vincristine, or who have had their hair permanently waved, then you will have heard about a treatment targeting the cytoskeleton of the cell. The Irish potato famine of 1850 had its origins in a component of the cytoskeleton and some viruses are transported to the cell nucleus along microtubules, a component of the cytoskeleton. There is some evidence that in Alzheimer’s disease microtubules in nerve axons are lost and that tangled protein increases. The cytoskeleton is a very important dynamic part of a cell, but it is not often shown in simplified drawings.
All cells, except those of most bacteria, contain components of the cytoskeleton. They help the cell remain rigid but also help it move and change its shape when instructed to do so. Components of the cytoskeleton also enable cilia, flagella and sperm to move, cell organelles to be moved and positioned, and muscles to function. During cell division these components also assist by pulling the daughter chromosomes to opposite ‘poles’ in the dividing process. Throughout the life of the cell various molecules and cargo containing vesicles are transported around the cell by motor proteins. These move along the protein filaments using them as trackways rather like a railway locomotive runs on rail tracks.
There are three groups of movers, the motor proteins: kinesin, dynein and myosin, and three main groups of shapers, the protein filaments: microtubules, intermediate filaments and actin filaments.
-
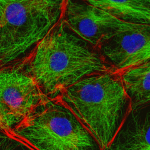
This image shows some animal cells. They are stained with fluorescent labels to help visualise the cytoskeleton with microtubules (green), actin filaments (red), and the nucleus (blue).
The cytoskeleton is not usually shown in simple diagrams of the cell because it is a complex meshwork of strands. Cells would not be cells with out their cytoskeleton (Images courtesy of Mark Shipman, James Blyth and Louise Cramer, Laboratory for Molecular Cell Biology, University College London, UK)
The cytoskeleton contributes to the architecture and transport system of the cell
In the early evolution of eukaryote cells the compartmentalisation of cell functions into membrane bounded structures, was accompanied by the evolution of a system that positioned and anchored them. This system therefore contributes to the architecture of the cell, its rigidity and in some cases to its ability to move. It also contributes by providing a physical transport system that enables cargo filled vesicles, some individual molecules, and even some cell organelles to be moved within the cell. The cytoskeleton is a dynamic entity with some cytoskeletal components being assembled and disassembled to meet the changing needs of the cell.
What are these movers and shapers in the cell?
Shapers (protein filaments) come in three sizes
The variable shape and rigidity of the cell and its ability to move is largely dependent on three groups of cytoskeletal protein filaments:
- Microtubules – size: about 25nm external diameter
- Intermediate filaments – size: about 10nm external diameter
- Actin filaments – size: about 8nm external diameter
All three groups of protein filaments are polymers made up of protein sub-units.
Movers (motor proteins) come in three models
Molecules and cargo containing vesicles, and sometimes organelles, are moved around the cell by motor proteins. There are three main groups of motor proteins, all powered in an efficient way by adenosine triphosphate (ATP)
- Kinesin
- Dynein
- Myosin
Like all the components of the cell, members of the cytoskeleton work in conjunction with other parts of the cell as a dynamic whole. For descriptive purposes the different parts will be discussed separately but they should be viewed, as they operate, as a total system.
Microtubules
Microtubules are hollow tubes of variable length and about 25nm diameter. They are stiff but flexible. Microtubules carry cargoes along the length of nerve axons; in humans axons these can be more than a meter in length.
Microtubules are assembled linearly from building blocks of tubulin molecules grouped into pairs called a dimer. Dimers are joined end-to-end by the process of polymerisation to form a linear polymer called a protofilament. Thirteen protofilaments lying in parallel are formed into a circular tube with the duct running down the middle. – This is a microtubule.
For microtubule assembly to take place the concentration of tubulin molecules in solution must exceed a critical level. Each tubulin building block molecule is said to be ‘polar’ since it has a different molecular configuration at each end. One end is called the ‘plus’ end; the other the ‘minus’ end. The ‘plus’ end of one molecule can only link to the ‘minus’ end of another. In this way they are stacked up to form a linear polymer.
Tubulin molecules also link with adjacent tubulin molecules in the circle rather as a circle of people could link up by holding hands.
Microtubules are very unstable and can disassemble very quickly. On first thoughts this would appear to be very inefficient but during mitosis and in circumstances that demand a rapid change of cell shape, quick disassembly and assembly are useful assets. This process is called ‘dynamic instability’ and is mainly directed and controlled by the chemical guanosine 5’ triphosphate (GTP).
Microtubule lengthening is brought about by the polymerisation of tubulin molecules; depolymerisation brings shortening.
Microtubule associated proteins (MAPs) in the cytoplasm regulate the lengthening and shortening process. They can bind to a microtubule at both its ends and along its length. In so doing MAPs can restrict lengthening and help attach microtubules to organelles and membranes within the cell.
Most microtubules are attached to, and initially arise from, an organising centre; in animal cells this is generally the centrosome. When cells divide the centrosome also divides. Microtubules are attached to the centrosome at their ‘minus’ end, the end that is slowest growing. Centrosomes often lie close to the cell nucleus and microtubules radiate from here in all directions towards the edge of the cell (plasma membrane).
The ‘plus’ end of the microtubule is furthest away from the centrosome. This is where microtubules rapidly lengthen or shorten in response to signals.
Microtubules in plant cells
Plant cells do not have a centrosome and hence no single observable nucleation site from which new microtubules are produced.
In plant cells there are many small nucleation sites and these and the microtubules they initiate are located in the cell on the cytoplasm side of the plasma membrane and just below it. They are aligned parallel to one another but are closely interlinked to form a network layer running parallel to the plasma membrane throughout the cell.
Intriguingly plant cell microtubules can re-align themselves in response to chemical stimulation. Microtubules in cells near the root tip are found at right angles to the direction of the growth of the root. Further back from the root tip the microtubules rotate through a right angle and align themselves to become parallel to the direction of root growth.
The importance of microtubules in plant cells should not be underestimated. Depolymerisation of microtubules causes cellulose to be laid down in a disorganised way. The root tip becomes a mass of such cells and although they expand they cannot elongate and the tissue grows in a distorted manner. Chemicals such as colchicine inhibit polymerisation and hence stop the production of microtubules. Some synthetic weedkillers bring about microtubule depolymerisation.
Microtubules and movement
The rapid assembly and disassembly of microtubules can physically move vesicles and cell organelles. Microtubules can locate them within the cell, hold them in position and re-locate them. This happens for example during mitosis.
Microtubules in Flagella and Cilia
In organisms such as Chlamydomonas and Paramecium the whiplike movements of flagella and cilia are created by microtubules working with motor proteins to produce bending movements by making microtubules slide. ATP, cAMP and calcium ions are also highly involved in movement in flagella and cilia.
Microtubules and protein (molecular) motors
Microtubules form the network of major trackways along which motor proteins travel. They travel along the outside of the microtubule NOT along the inside.
Actin filaments are also used as trackways and microtubules and actin filaments can be linked by proteins.
Motor proteins of different models travel along microtubules in different directions.
Kinesin models of motor protein travel toward the ‘plus’ end of a microtubule and tend to transport cargoes away from the area of the nucleus.
Dynein models travel toward the ‘minus’ end and hence transport cargoes along a microtubule to the nucleus.
Microtubules and Medicine.
Alzheimer’s disease
Microtubules found in nerve cells are attached to microtubule associated proteins (MAPs) including tau protein. In healthy cells these proteins are attached to the outside of microtubules. As well as regulating them (see above) MAPs increase both the stability and stiffness of the microtubules, but reduce their flexibility.
In Alzheimer’s disease and some other dementias microtubules are lost from the axons and there is an increase in unorganised tangled filaments of tau protein.
The loss of microtubules results in the loss of a transport service up and down the nerve axon. Without this movement of biochemicals and organelles the nerve cell looses function. The causes of microtubule loss and the increase in tau are not yet known and the total picture is thought to be more complicated.
Microtubules and cancer treatment
Any drug regime or intervention procedure that inhibits the physical or chemical processes/ events that are part of the process of cell replication has potential as a treatment for cancer. To be worthy of consideration however the drug or procedure must not only to be effective but have minimal or no disruptive effects on non-cancerous cells. It must also be capable of being delivered to the target cells in an efficient and safe way with few or no general side effects such as nausea.
Microtubules play such an important role during M-phase of the cell cycle that they provide a possible target for cancer treatment.
Consider what microtubules in the form of spindle fibres do during mitosis. They align then pull and position. This is performed in the main by both the growing and shortening of microtubules. If this mechanism can be stopped, then the cell will be prevented from dividing. The cell biology indicates two possible options:
- disrupting microtubule assembly. – This means preventing polymerization and/or promoting depolymerisation, and
- ‘locking’ or stabilizing microtubules already assembled so they cannot disassemble by depolymerising.
Several drugs have been found to fit the cell biology.
Taxol works by inhibiting microtubular disassembly. The cell becomes ‘locked’ with microtubules becoming static – not a situation conducive to cancer cell division.
The drugs colchicine, nocodazole and vincristine inhibit microtubule assembly by inhibiting polymerisation and promoting depolymerisation. Vincristine particularly has been used in cancer chemotherapy.
Work in progress
Please return to this site to see notes about intermediate and actin filaments and motor proteins. – These items are forthcoming.